Scientists have created the first-ever molecular catalyst to create solid-state reactions, enabling high-efficiency transformations at near room temperature.
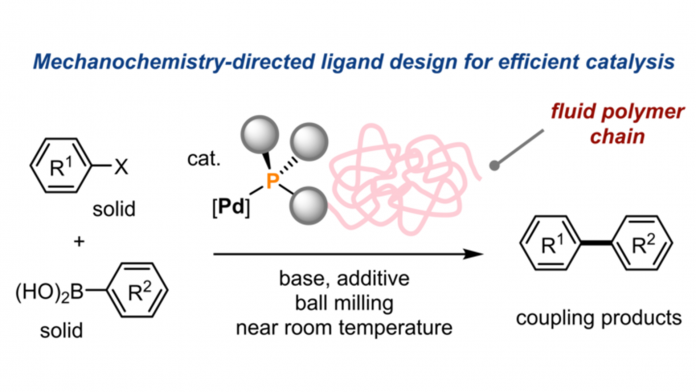
The chemists at Hokkaido University and the Institute for Chemical Reaction Design and Discovery (WPI-ICReDD) found that attaching long polymer molecules to a metal catalyst could trap the catalyst in a fluid phase, which enabled efficient solid-state reactions at near room temperature.
The study, ‘Mechanochemistry-Directed Ligand Design: Development of a High-Performance Phosphine Ligand for Palladium-Catalyzed Mechanochemical Organoboron Cross-Coupling,’ was reported in the Journal of the American Chemical Society. If this process was to be adapted for broad application in the field of chemical research and industry, it could bring significant cost and energy savings.
Solid-state reactions vs solution-based reactions
Chemical synthetic reactions are usually performed in solution, where dissolved molecules can intermingle and react freely.
However, in recent years, chemists have developed a process called mechanochemical synthesis, in which solid-state crystals and powders are ground together. This approach typically works better because it reduces hazardous solvents and allows reactions to proceed faster and at lower temperatures, saving energy costs. Moreover, it can be used for solid-state reactions between compounds that are difficult to dissolve in available solvents.
Solid-state reactions occur in a very different environment than solution-based reactions. Previous studies found that palladium complex catalysts initially designed for use in solution often did not work sufficiently in solid-state mechanochemical reactions and that high reaction temperatures were required.
Using the unmodified palladium catalyst for solid-state reactions resulted in limited efficiency due to the tendency of palladium to aggregate into an inactive state. The team chose a new direction, designing a catalyst to overcome this mechanochemical problem of aggregation.
“We developed an innovative solution, linking palladium through a specially designed phosphine ligand to a large polymer molecule called polyethene glycol,” explained Professor Hajime Ito, co-author of the study.

The best-performing catalyst to date
The polyethene glycol molecules form a region between the solid materials that behave like a molecular-level fluid phase, where mechanochemical Suzuki-Miyaura cross-coupling reactions proceed much more efficiently and without the problematic aggregation of palladium.
In addition to achieving significantly higher product yields, the solid-state reaction proceeded effectively near room temperature – the previously best-performing alternative required heating to 120°C. Similar cross-coupling reactions are widely used in research and the chemical industry.
“This is the first demonstration of a system that is specifically modified to harness the potential of palladium complex catalysts in the unique environment of a mechanochemical reaction,” said Associate Professor Koji Kubota, who conducted the study alongside Ito.
They believe it could be adapted for many other reactions, as well as for catalysts using different elements from the transition metals of the periodic table.
The wider adoption of the process, and others like it, could bring significant savings in costs and energy consumption in commercial chemical processes while allowing more environmentally friendly large-scale production of many beneficial chemicals. The study is revolutionary in the field of solid-state reactions.