Dr Warren L Grayson outlines the work taking place at his lab at the Johns Hopkins’ Translational Tissue Engineering Center in regenerating musculoskeletal tissues, from treating skeletal muscle loss to 3D-printing bioactive scaffolds with precise anatomic geometries
Damage to tissues of the musculoskeletal system – i.e. bone, muscle, cartilage, ligaments, and tendons – are among the most common injuries that occur during everyday activities. They are seldom life threatening, and many of these injuries heal naturally following rest and temporary disuse. Hence, surgical interventions are rare. Yet, when damage to these tissues overwhelms the body’s natural regenerative responses, they almost always require surgical interventions. In the worst cases, where traumatic injury results in volumetric tissue loss, there are no great surgical solutions. The current state-of-the-art treatment for replacing a missing portion of facial bone or a section of the quadriceps muscle is to ‘rob Peter to pay Paul’; that is, take similar tissue from another part of the patient’s body and use it to repair the damaged section.
To overcome this limitation, in the late 1980s the idea of blending biomaterials with cells to generate ‘living tissues’ was successfully launched and the field of tissue engineering – the creation of biological prostheses – was born. Since then, there have been numerous reports of bio-engineered organs and tissues ranging from skin to mini-brains. The strategies have evolved over the ensuing three decades to include organoids and 3D bio-printing along with advances in biomaterial designs. Consequently, the field is now capable of engineering complex musculoskeletal tissues comprised of multiple cell types with the appropriate spatial organisation to facilitate functional outcomes. Yet, bottlenecks remain in the translation of these strategies into successful clinical therapies or breakthrough commercial products. This includes novel bone scaffolds for treating facial injuries, which is the primary focus of the Grayson Laboratory for Craniofacial and Orthopaedic Tissue Engineering.
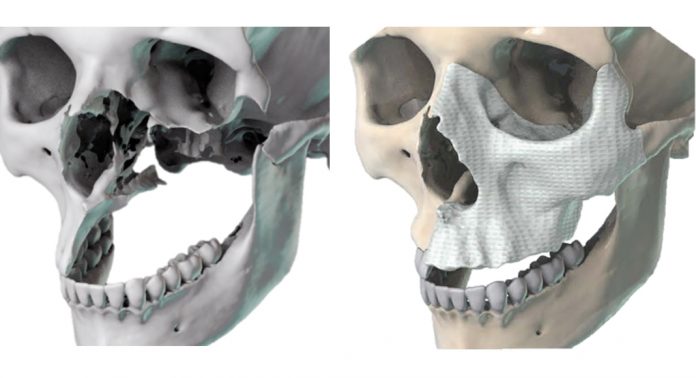
Each year, there are approximately 200,000 surgeries in the United States to resolve facial bone loss that require bone transplantation. These defects are large and, due to the irregular contours of facial bone, geometrically complex. Personalised tissue engineering strategies have the potential to treat these defects by combining the patient’s own stem cells with porous, biodegradable scaffolds and biological cues to regenerate missing bone. However, traditional tissue engineering approaches face practical and logistical barriers associated with the need to grow tissues ex vivo prior to transplantation into the patient. The development of a clinical point-of-care tissue-engineering strategy has the potential to optimise the regeneration of bone tissues and would revolutionise the therapeutic landscape for patients.
3D printing biodegradable scaffolds to promote new bone growth
Facial bone defects are difficult to treat due to their patient- and defect-specific anatomic geometries that impact aesthetic and functional outcomes. Metallic implants have sub-optimal outcomes owing to their excessive material strength, poor integration within native bone, and high risk of infection. Bone cement implants have also been used but have considerable disadvantages.
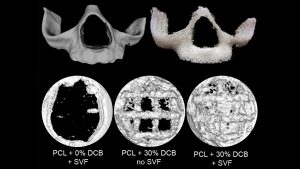
Alternatively, biodegradable polymer scaffolds allow for the sustained restoration of facial form and function and facilitate the regeneration of living bone tissue, which can dynamically adapt to its environment. 3D-printable thermoplastic polymers have sufficiently high print resolution, exhibit excellent biocompatibility, and have controllable biodegradation rates. The bioactivity of synthetic scaffolds is enhanced by a decellularised bone matrix without a significant reduction in the mechanical properties of the scaffolds.
The Grayson Lab 3D-prints bioactive scaffolds with precise anatomic geometries and has tested their bone regeneration capacity in models of traumatic bone loss in the skulls of mice. Generating customised, bioactive materials that can be aseptically prepared and provided to the surgeon ahead of surgery for point-of-care treatments have the potential to radically transform treatment for patients.
Fat-derived stem cells for bone repair
Multiple cell types may be encapsulated into the scaffolds to promote new bone growth. Our team has a strong record of collaborative publications using various stem cell populations. However, our major interests lie in utilising the cells that can be isolated from lipoaspirated fat and used intraoperatively without the need for in vitro cell expansion.
A typical isolate may contain upwards of 500,000,000 cells from a standard non-obese patient. The isolate provides progenitor cells capable of regenerating new blood vessels as well as new bone. This incredible potential combined with the abundance, ease of procurement, and potential to be used as a point-of-care therapy affords fat-derived cells a unique advantage that we harness for facial bone regeneration.
Studies in our lab have demonstrated the incredible potential of combining fat-derived stem cells with 3D-printed scaffolds in treating bone loss in mice. Ongoing studies in the Grayson Lab continue to examine their potential in large pre-clinical models and, in the not-too-distant future, we plan to test the capacity of this strategy in humans.
Volumetric muscle loss
The Grayson Lab also tackles the challenging problem of treating skeletal muscle loss. Skeletal muscle makes up almost half of the human lean body mass and approximately 40% of all traumatic injuries involve skeletal muscle damage. While skeletal muscle possesses an intrinsic self-repair and regeneration capacity, cases of volumetric muscle loss (VML) lead to chronic functional deficits in the affected muscle. This results in a significant decrease in motor function and impaired quality of life.
One major cause of VML is trauma. Currently, there are no effective therapies available to replace or effectively regenerate the lost muscle to overcome the reduced contractility and limited function. Our lab has developed methods to engineer functional skeletal muscle grafts using biomaterials that mimic the stiffness and structure of native muscle while promoting tissue assembly in vivo and maximising vascular and neural ingrowth.
Electrospun fibrin hydrogels
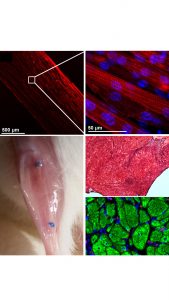
In addition to the autografts used to treat VML, decellularised extracellular matrix (dECM) scaffolds have been used pre-clinically and clinically. The dECM scaffolds – often used without the addition of cells – improve muscle function through the development of scar tissue and minimal new muscle growth. Conversely, synthetic, biodegradable polymer matrices are tuneable but do not provide biologic cues.
Through highly collaborative approaches with material scientists we have employed electrospun hydrogels (our team used fibrin – a naturally occurring polymer in the blood) to guide the alignment and assembly of mouse muscle cells into contractile muscle tissues that sustained the restoration of skeletal muscle form and function after being used to treat VML.
Advanced bioreactor systems for tissue maturation, preservation, and testing
The unique physical environments to which cells and tissues are exposed govern their behaviour. Musculoskeletal tissues in particular exert or resist mechanical forces. Mimicking the native physiology guides stem cell differentiation and tissue development. Bioreactor systems are central to the application of ‘biophysical’ cues and our team has extensive experience in designing, building, and testing advanced bioreactor systems capable of regulating multiple stimuli simultaneously.
For example, custom-designed ‘multi-parametric bioreactor systems’ apply mechanical forces (e.g. stretching or compression), electrical signals, and enhanced oxygen delivery by using flow perfusion to mature the engineered tissues, assess their functionality in a non-destructive manner, or preserve the function of native tissues used for transplantation.
Collaborative and Translational Tissue Engineering
The Grayson Lab is housed in Johns Hopkins’ Translational Tissue Engineering Center. The centre comprises 10 core faculty members with expertise covering drug delivery, biomaterial design, protein and immune engineering, glycoengineering, and transplant surgery. The laboratories are designed with an open floor plan which promotes interdisciplinary and collaborative research. The centre is committed to translating its research to improve the lives of patients. Consequently, our labs participate in clinical trials and industry collaborations. There are several start-up companies emanating from our centre and built around a spectrum of enabling technologies for advancing regenerative medicine in nearly every clinical field.
References
1 Temple JP, Hutton DL, Hung BP, Huri PY, Cook CA, Kondragunta R, Jia XF, Grayson WL, ‘Engineering Anatomically Shaped Vascularized Bone Grafts with hASCs and 3D-printed Bone Scaffolds’. J Biomedical Materials Research, Part A. 2014, 102(12): 4317 – 4325
2 Nyberg EL, Farris AL, O’Sullivan A, Rodriguez R, Grayson WL, ‘Comparison of SVF and Passaged ASCs as Point-of-Care Agents for Bone Regeneration’. Tissue Engineering Pt. A, 2019, 25(21-22): 1459-1469
3 Gilbert-Honick J, Iyer SR, Somers SM, Lovering RM, Wagner K, Mao HQ, Grayson WL,’ Engineering functional and histological regeneration of vascularized skeletal muscle’. Biomaterials, 2018, 164:70-79
4 Gilbert-Honick J, Iyer SR, Somers SM, Takasuka H, Lovering RM, Wagner K, Mao HQ, Grayson WL, ‘Engineering 3D skeletal muscle primed for neuromuscular regeneration following volumetric muscle loss’. Biomaterials, 2020, Accepted
Dr Warren L Grayson
Professor of Biomedical Engineering
Johns Hopkins University School of Medicine
+1 410 502 6306
wgrayson@jhmi.edu
Tweet @GraysonLab
https://pages.jh.edu/~graysonlab
Please note, this article will also appear in the fourth edition of our new quarterly publication.